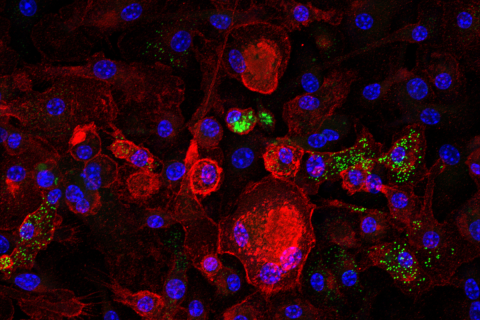
In a mouse and human study, a UCLA-led team has found how an enzyme called ULK1, working in the liver, protects against metabolic dysfunction associated fatty liver disease (MASLD), an obesity-linked liver condition that is a leading cause of liver failure requiring a transplant.
The findings of the study, published April 2 in the peer-reviewed Journal of Clinical Investigation, unlock previously unrecognized insights about ULK1’s function that open a path to new therapies for MASLD. Dr. E. Dale Abel, chair of the UCLA Department of Medicine, and Young Do Koo, assistant professor in the division of endocrinology, diabetes and metabolism at the David Geffen School of Medicine at UCLA, led the research team.
MASLD is a highly variable syndrome that in some people can progress to metabolic dysfunction-associated steatohepatitis (MASH), an advanced form of fatty liver disease that can lead to tissue scarring, cirrhosis, liver failure and liver cancer. MASLD is closely linked to insulin resistance and obesity-associated chronic disease, and its incidence is rapidly rising worldwide. However, the mechanisms that lead to its development and progression remain poorly understood.
Thus, while treatments like GLP-1 agonists that mitigate some of the features of MASLD may act in part by reducing obesity, there are few therapies specific to the condition itself. However, some evidence suggests that autophagy — a naturally-induced state in which the body clears out, recycles or repairs old or dysfunctional cells in response to nutrient deprivation or fasting — can protect against MASLD. There is therefore much interest in leveraging autophagy as a therapeutic strategy.
The study by Abel’s team adds nuance to this concept and reveals that ULK1 could be an important therapeutic target. ULK1 has been well-studied for its role in autophagy, but it was previously unclear whether autophagy alone could be responsible for any role that ULK1 could have in protection against MASLD. To establish a potential link, Dr. Abel’s team first analyzed data on the expression of ULK1 in liver samples from people with and without MASLD and in mice that were fed a high-fat diet (HFD). ULK1 expression was lower in people with the disease compared to healthy individuals, indicating a correlation between MASLD and ULK1 deficiency.
Next, the scientists developed a mouse model lacking the gene that encodes for ULK1 specifically in liver cells — in other words, a liver- specific ULK1 “knockout” model. A series of experiments with the ULK1 knockout mouse model demonstrated that it plays a direct, independent role in MASLD development: Mice without the protein in their livers had higher levels of liver triglycerides and progressed to MASH when fed a HFD. They also more readily developed insulin resistance and glucose intolerance. Even in animals that had not yet become obese or developed insulin resistance, the absence of the gene sped up fatty acid and triglyceride creation in the liver.
Unexpectedly autophagy was not impaired in the liver even in the absence of ULK1, suggesting that the mechanism by which ULK1 protects from the development of MASLD are independent from its role in autophagy. ULK1 is a specific type of enzyme called a kinase, which exerts its activity on cellular processes via phosphorylation, which means attaching a phosphate group to a protein in order to switch its activity on or off. In this case, the researchers hypothesized that ULK1 might be influencing fat synthesis in the liver by phosphorylating proteins with direct roles in that process.
Their experiments pointed to a protein called NCOA3, which regulates fat synthesis through its interactions within a multi-protein complex in the nucleus called NCOA3-CBP-CREB. The team found that phosphorylation of NCOA3 by ULK1 prevents fat synthesis in the liver, and that knocking out the genes for both ULK1 and NCOA3 in the same mouse, prevented the MASLD and insulin resistance observed in the mice in which only ULK1 was knocked out. This NCOA3-dependent effect was validated in experiments where NCOA3 was inhibited with a small molecule, SI-2. Treatment of ULK1 KO mice with SI-2 normalized fat synthesis in the livers of these animals despite the fact that they did not possess the ULK1 gene, demonstrating that its interaction with NCOA3 was the culprit behind MASLD development. NCOA3 inhibition with SI-2 also reduced inflammation in livers of ULK1 knockout mice, suggesting that it works at multiple levels against the mechanisms that lead to MASLD. Changes in these inflammatory pathways were also noted in human samples from MASLD patients who had reduced levels of ULK1.
There is still much more to learn about how ULK1 comes to be repressed in MASLD in the first place. Future studies by the same team will seek to elucidate these mechanisms. In the meantime, they will continue to explore the development of drugs that target the interaction between ULK1 and NCOA3, with the aim of ultimately testing them in humans.
In addition to Dr. Koo and Dr. Abel, UCLA scientists involved with the study are affiliated with the Vatche and Tamar Manoukian Division of Digestive Diseases at the UCLA David Geffen School of Medicine, the UCLA Comprehensive Liver Research Center, the UCLA Department of Microbiology, Immunology and Molecular Genetics, the UCLA Institute for Quantitative and Computational Biosciences, and the UCLA Department of Molecular and Medical Pharmacology. The team also included researchers affiliated with the Veterans’ Affairs Greater Los Angeles Healthcare System, the Vanderbilt University Department of Molecular Physiology and Biophysics, and the University of Iowa Carver College of Medicine Fraternal Order of Eagles Diabetes Research Center and Department of Anatomy and Cell Biology.
The study was funded by the American Heart Association Strategically Focused Research Network.